Background: MCL is an aggressive non-Hodgkin lymphoma (NHL) subtype which remains incurable despite improvements in progression free survival with consolidative autologous transplant and the introduction of novel targeted therapies [Dreyling et al. Blood 2012]. CD19-directed chimeric antigen receptor (CAR) T-cell therapy (CD19-CAR) has shown durable disease control in r/r MCL, but current standard of care products are limited by high rates of grade 3+ immune-related toxicities [Wang et al. NEJM 2020]. We hypothesized that combining a naïve/stem-memory (T N/SCM) phenotype-enriched CD19-CAR T-cell product with the selective BTK inhibitor (BTKi) acalabrutinib would attenuate the rates of severe toxicities, improve durable remission rates, and obviate the need for indefinite use of acalabrutinib in r/r MCL.
Methods: We conducted a phase 1 single center, prospective clinical trial (NCT04484012) evaluating a single infusion of T N/SCM-enriched CD19.28.z.EGFRt-CAR at a target dose of either 2x10 8 (DL1) or 5x10 8 (DL2) CAR+ cells (≤20% lower dose allowed) in combination with ongoing acalabrutinib 100 mg twice daily for patients with r/r MCL after at least 1 prior line of therapy. Patients were required to undergo at least 3 months but less than 7 months of treatment with a BTKi prior to enrollment without evidence of progression and with measurable disease per Lugano criteria. All patients were switched to acalabrutinib for protocol therapy prior to lymphodepletion, and planned to continue until day 180 post CD19-CAR infusion at which time all therapy would be discontinued. We report outcomes from the dose finding portion of the study. Descriptive statistics were utilized for baseline characteristics and survival analyses were calculated utilizing the Kaplan-Meier method. Investigator-assessed response rates were based on Lugano criteria. Minimal residual disease (MRD) status was determined using either multiparameter flow cytometry (FCM) or next-generation sequencing (NGS).
Results: To date, 8 patients enrolled and have received CD19-CAR (n=4 at DL1; n=4 at DL2). Manufacturing was successful in 100% of patients. Median time from leukapheresis to product infusion was 53 days (range, 41-114). Treated patients were 75% male; with a median age of 63 years (range, 38-70); and median number of prior therapies of 2 (range, 2-3). All patients had bone marrow involvement, and had high risk features including progression of disease within 24 months (n=6), relapse after autologous HCT (n=2), TP53 mutations (n=6), Ki-67 ≥30% (n=4), and/or complex karyotype (n=1). Prior to enrollment, all patients had received BTKi (n=8 acalabrutinib) for a median of 6.1 months (range, 4.5-7.3). No BTK and PLC-γ mutations were detected in patients with available samples.
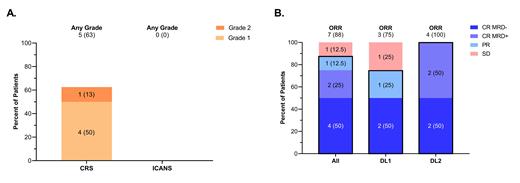
The combination of acalabrutinib and CD19-CAR was well tolerated; 5 patients (63%; n=1 grade 2; n=4 grade 1) had cytokine release syndrome (CRS), with no severe cases. No patients (0%) had immune effector cell associated neurotoxicity syndrome (ICANS) [Figure 1A]. No patient required corticosteroids for toxicity management, and only 2 patients (n=2/5, 40%) required tocilizumab for CRS. There were no serious adverse events (SAEs), and no treatment discontinuations due to AEs observed. The best ORR was 88% [n=6 CR (75%); n=1 PR (13%)], with median time to best response of 28 days. In those 6 patients who achieved CR, 2 patients were FCM-MRD negative (<10 -4) and 2 were NGS-MRD negative (<10 -6) (n=4/6, 67%) [Figure 1B]. After a median follow-up of 18.5 months (range, 8.3-28.9), 5 of 6 patients remain in CR, while1 patient who achieved an initial CR relapsed at 9.2 months. At 1-year post-infusion, the cumulative incidence of relapse and NRM were 30% (95% CI, 3%-67%) and 0%, respectively. The 1-year PFS and OS were 70% (95% CI: 22%-92%) and 100%, respectively.
Conclusion: The combination of CD19-CAR with acalabrutinib was well tolerated and effective, yielding an ORR 88% and MRD- rate 50% in a cohort of patients with high-risk MCL. Notably, CRS events were manageable and low grade, and no ICANS was observed. Enrollment is ongoing in the expansion cohort (DL2), and additional clinical and correlative analyses will be presented at the meeting.
OffLabel Disclosure:
Baird:Kite Pharma-Gilead: Research Funding, Speakers Bureau; Genentech-Roche: Research Funding; Regeneron Pharmaceuticals: Research Funding; Cellular Biomedicine Group: Research Funding. Shouse:Beigene, Inc.: Speakers Bureau; Kite Pharmaceuticals: Consultancy, Speakers Bureau. Danilov:Genentech: Consultancy; Janssen: Consultancy; Beigene: Consultancy, Research Funding; Bayer: Research Funding; Abbvie: Consultancy, Research Funding; Nurix: Consultancy, Research Funding; Merck: Consultancy; GenMab: Consultancy, Research Funding; Astra Zeneca: Consultancy, Research Funding; Bristol Meyers Squibb: Consultancy, Research Funding; Lilly Oncology: Consultancy, Research Funding; MEI: Consultancy, Research Funding; Cyclacel: Research Funding. Herrera:Adicet Bio: Consultancy; Caribou Biosciences: Consultancy; Karyopharm Therapeutics: Consultancy; AstraZeneca/MedImmune: Consultancy; Pfizer: Consultancy; BMS: Consultancy, Other: Travel/Accommodations/Expenses, Research Funding; Kite, a Gilead Company: Research Funding; Tubulis GmbH: Consultancy; ADC Therapeutics: Consultancy, Research Funding; Genentech/Roche: Consultancy, Research Funding; Takeda: Consultancy; Genmab: Consultancy; Regeneron: Consultancy; Merck: Consultancy, Research Funding; Allogene Therapeutics: Consultancy; AbbVie: Consultancy; Seattle Genetics: Consultancy, Research Funding; Gilead Sciences: Research Funding; AstraZeneca: Research Funding. Popplewell:La Roche: Honoraria; Hoffmann: Honoraria; Seattle Genetics: Consultancy, Honoraria; Novartis: Consultancy; Pfizer: Honoraria. Rosen:Pheromone Bio, Inc: Consultancy; Exicure: Consultancy; Apobiologix/Apotex Inc: Consultancy; PharmaGene, LLC: Consultancy; Trillium Therapeutics, Inc: Consultancy; Verastem, Inc: Consultancy; NeoGenomics: Membership on an entity's Board of Directors or advisory committees; Pepromene Bio, Inc: Membership on an entity's Board of Directors or advisory committees; Pepromene Bio, Inc: Current holder of stock options in a privately-held company; Exicure: Current holder of stock options in a privately-held company; January Biotech: Current holder of stock options in a privately-held company; Trillium Therapeutics: Current holder of stock options in a privately-held company; SLAM BIOTHERAPEUTICS, INC: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees. Kwak:PeproMene Bio. Inc: Consultancy, Current equity holder in private company. Budde:Amgen: Research Funding; ADC Therapeutics: Consultancy; AstraZeneca: Consultancy, Research Funding; MustangBio: Research Funding; Roche: Consultancy; Novartis, Gilead, F. Hoffmann-La Roche Ltd, BeiGene, Genentech, Inc.: Consultancy; Merck: Research Funding.
CD19.28.z.EFGRt CAR-T cells combined with acalabrutinib for the treatment of relapsed or refractory mantle cell lymphoma.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal